TESTING FUELS -- CHEMICAL REACTIONS
Assignment #4: Acids vs. Bases -- pH Scale
1) Read the following information:
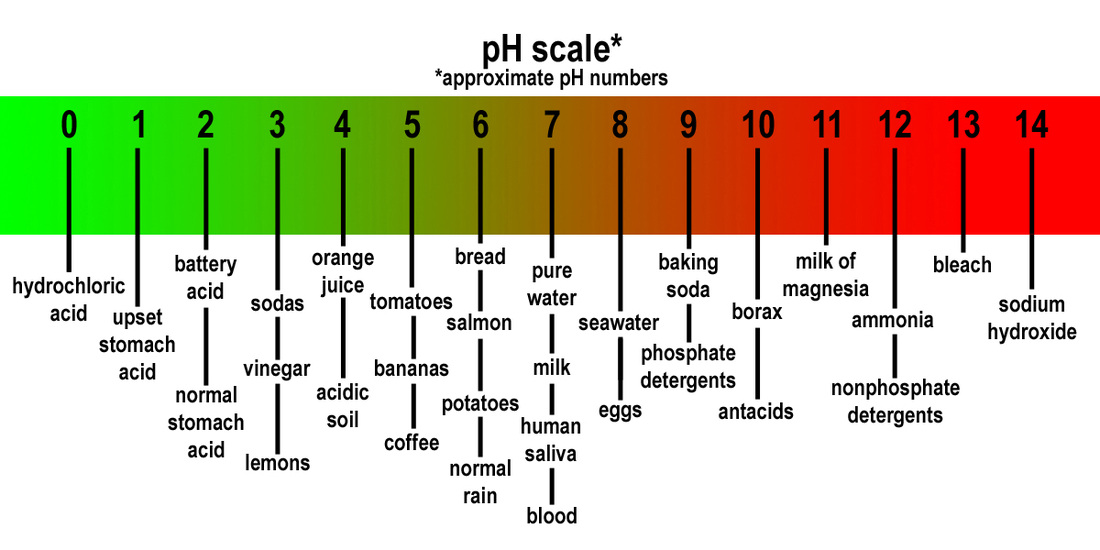
~ Acids are ionic compounds (a compound with a positive or negative charge) that break apart in water to form a hydrogen ion (H+).
.. The strength of an acid is based on the concentration of H+ ions in the solution. The more H+ the stronger the acid. Example: HCl (Hydrochloric acid) in water
.. CHARACTERISTICS of acids:
1) Acids taste sour
2) Acids react strongly with metals (Zn + HCl)
3) Strong Acids are dangerous and can burn your skin
~ Bases are ionic compounds that break apart to form a negatively charged hydroxide ion (OH-) in water.
.. The strength of a base is determined by the concentration of Hydroxide ions (OH-). The greater the concentration of OH- ions the stronger the base. Example: NaOH (Sodium Hydroxide-a strong base) in water
.. CHARACTERISTICS of bases:
1) Bases taste bitter
2) Bases feel slippery
3) Strong bases are very dangerous and can burn your skin
2) Watch following video on "Importance of pH in every day life" (below).
~ Acids are ionic compounds (a compound with a positive or negative charge) that break apart in water to form a hydrogen ion (H+).
.. The strength of an acid is based on the concentration of H+ ions in the solution. The more H+ the stronger the acid. Example: HCl (Hydrochloric acid) in water
.. CHARACTERISTICS of acids:
1) Acids taste sour
2) Acids react strongly with metals (Zn + HCl)
3) Strong Acids are dangerous and can burn your skin
~ Bases are ionic compounds that break apart to form a negatively charged hydroxide ion (OH-) in water.
.. The strength of a base is determined by the concentration of Hydroxide ions (OH-). The greater the concentration of OH- ions the stronger the base. Example: NaOH (Sodium Hydroxide-a strong base) in water
.. CHARACTERISTICS of bases:
1) Bases taste bitter
2) Bases feel slippery
3) Strong bases are very dangerous and can burn your skin
2) Watch following video on "Importance of pH in every day life" (below).